Access deep patient data
Richly described multimodal patient data to meet your needs




Owkin’s data is different
We provide high-quality, context-rich patient data that can be queried to understand it the way you need. Our direct partnerships with 100+ leading hospitals in 14 countries give us unparalleled deep data access - made possible through 9+ years of close engagement with clinicians and PIs.
This allows us to collect cutting-edge data modalities along with rich clinical characterization to build unmatched descriptions of each patient.
Title here please
- Non Small-Cell Lung Cancer
- Breast Cancer
- Bladder Cancer
- Ovarian Cancer
- Glioblastoma
- Diffuse Large B-Cell Lymphoma
- Colorectal Cancer
- Head & Neck Cancer
- Pancreatic Cancer
- Mesothelioma
- Gastric Cancer
- Irritable Bowel Disease
- Pancancer (dx)
- Rheumatoid Arthritis
- Clinical data
- H&E (+Whole Slide Image)
- Bulk RNA sequencing
- Whole Exome Sequencing
- Single Cell RNA sequencing
- Spatial Transcriptomics
- Histological subtype
- Patient cohort
- Geographic origin
- Median follow up time
- Median age and age range
- Stage distribution
- Line of therapy repartition
- University Hospital Lille
(Lille, France – Academic Partner); - Technical University Munich
(Munich, Germany – Academic Partner); - Bern University Hospital, University of Bern
(Bern, Switzerland – Academic Partner); - Ramón y Cajal University hospital
(Madrid, Spain - Academic Partner); - University Hospital Basel
(Basel, Switzerland – Academic Partner); - Cardiff University
(Cardiff, Wales - Academic Partner)
- Under article 6 (GDPR): legitimate interest of Owkin to perform its research projects to develop, improve or validate its AI tools for better patient’s medical care or treatments (a LIA has been performed to assess the balance between the benefits for Owkin and risk for the patients);
- Under article 9 (GDPR): Scientific research purpose.
Compliance with MR-004 under French Law.
- Crohn’s & Colitis Foundation (New York, USA – Academic Partner);
- University of North Carolina (Chapel Hill, North Carolina, USA – Academic Partner);
- Finnish Biobank Cooperative - Helsinki Biobank and Tampere Biobank (Finland – Academic Partner).
- Under article 6 (GDPR): legitimate interest of Owkin to perform its research projects to develop, improve or validate its AI tools for better patient’s medical care or treatments (a LIA has been performed to assess the balance between the benefits for Owkin and risk for the patients);
- Under article 9 (GDPR): scientific research purpose
- Compliance with MR-004
- Gustave Roussy (Paris, France - Academic Partner);
- Erlangen University Hospital (Erlangen, Germany – Academic Partner);
- Charité University Hospital (Berlin, Germany – Academic Partner);
- Centre Hospitalier Universitaire Vaudois (Lausanne, Switzerland – Academic Partner);
- University of Pittsburgh (Pittsburgh, Pennsylvania, USA – Academic Partner).
- Under article 6 (GDPR): legitimate interest of Owkin to perform its research projects to develop, improve or validate its AI tools for better patient’s medical care or treatments (a LIA has been performed to assess the balance between the benefits for Owkin and risk for the patients);
- Under article 9 (GDPR): public interest in public health as the study will ensure high standards of quality and safety of health care.
- Under French Law: Compliance with MR-003
- Het Ziekenhuisnetwerk Antwerpen vzw (Antwerp – Belgium - Academic Partner);
- Gasthuiszusters Antwerpen vzw (Antwerp – Belgium - Academic Partner);
- Assistance Publique - Hôpitaux de Paris (Paris – France - Academic Partner);
- Gustave Roussy (Paris – France - Academic Partner).
- Aigora GmbH (München – Germany - Industrial Partner);
- Baylor Scott & White Health (Dallas, Texas – United States of America - Academic Partner);
- Cooper Health (Camden, New Jersey – United States of America - Academic Partner);
- Vall d'Hebron Research Institute (VHIR) (Barcelona – Spain - Academic Partner).
- University Hospitals Birmingham NHS Foundation Trust (Birmingham – United Kingdom - Academic Partner);
- CureCollect Ltd (London – United Kingdom - Academic Partner).
- Under article 6 (GDPR): legitimate interest of Owkin to perform its research projects to develop, improve or validate its AI tools for better patient’s medical care or treatments (a LIA has been performed to assess the balance between the benefits for Owkin and risk for the patients);
- Under article 9 (GDPR): scientific research purpose.
- Compliance with MR-004
- Assistance Publique - Hôpitaux de Paris (based in Paris- French Academic Partner)
- Hospices Civiles de Lyon (based in Lyon - French Academic Partner)
- Under Article 6 (GDPR): Legitimate interest of Owkin to to develop and improve artificial intelligence tools enabling better medical treatment (a legitimate interest assessment has been performed to assess the balance between the benefits for Owkin and risk for the patients)
- Under Article 9 (GDPR): Scientific research purposes
- Under French law: compliance with MR004
- Under Article 6 (GDPR): legitimate interest of Owkin to perform its research projects to develop, improve or validate its AI tools for better patients medical care or treatments (a LIA has been performed to assess the balance between the benefits for Owkin and risk for the patients)
- Under Article 9 (GDPR): the public interest in public health as the study will ensure high standards of quality and safety of health care and medical devices
- Under French law: compliance with MR004
- Institut Claudius Regaud (based in Toulouse - French Academic Partner)
- Gustave Roussy (based in Paris - French Academic Partner)
- Under Article 6 (GDPR): legitimate interest of Owkin to perform its research projects to develop, improve or validate its AI tools for better patients medical care or treatments (a LIA has been performed to assess the balance between the benefits for Owkin and risk for the patients)
- Under Article 9 (GDPR): the public interest in public health as the study will ensure high standards of quality and safety of health care and medical devices
- Under French law: compliance with MR004
- Unicancer (based in Paris - French Academic Partner)
- Institut Curie (based in Paris - French Academic Partner)
- Cypath-RB (based in Villeurbanne - French Non-Academic Partner)
- Under Article 6 (GDPR): legitimate interest of Owkin to perform its research projects to develop, improve or validate its AI tools for better patients medical care or treatments (a LIA has been performed to assess the balance between the benefits for Owkin and risk for the patients)
- Under Article 9 (GDPR): the public interest in public health as the study will ensure high standards of quality and safety of health care and medical devices
- Under French law: compliance with MR004
Patient Validation Hub

Start with patient data
The foundation of all our models is patient data. This keeps their initial predictions closer to the ground truth of biology and allows them to see patterns and activity that simpler systems miss.
That’s why our philosophy is always: from real-world patient data to real-world impact.


We work with our sister company, Waiv (formerly Owkin Dx), to bring AI models into diagnostic practice. Waiv has multiple AI products in the clinic, including RlapsRisk BC and the MSIntuit Suite, and co-development programs with MSD and AstraZeneca.
The AI behind these models was originally developed by Owkin, and has been through rigorous patient testing, earning the products high-level regulatory approval through CE-marking.
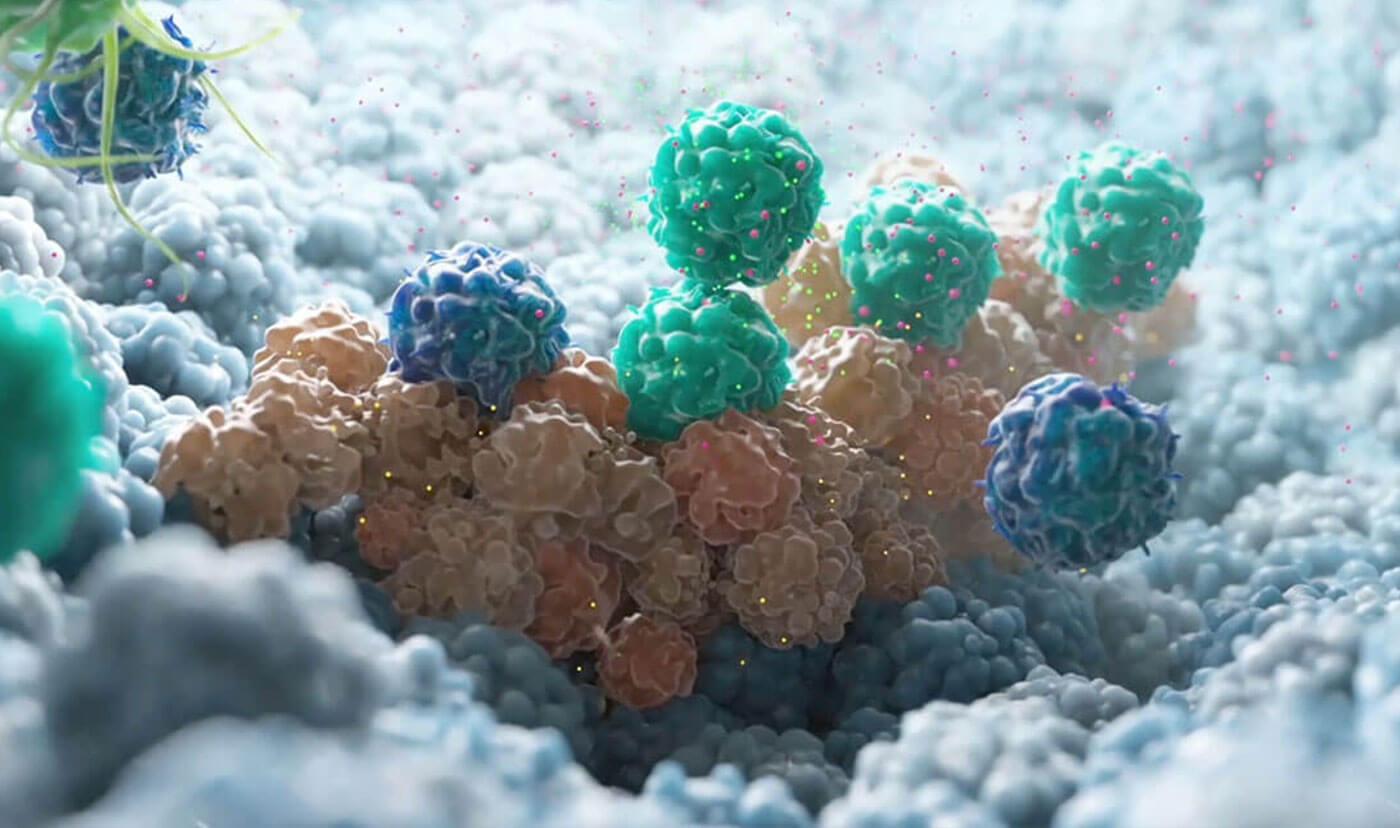

Clinical trials are the true test for any AI-proposed treatment. We run INVOKE - the clinical trial for our AI-enabled asset OKN4395 - testing the accuracy of our AI’s predictions in real world patients.
The trial regularly produces patient data which we continuously integrate into our ongoing AI training, to keep our AI based on real-world, up-to-date patient responses.
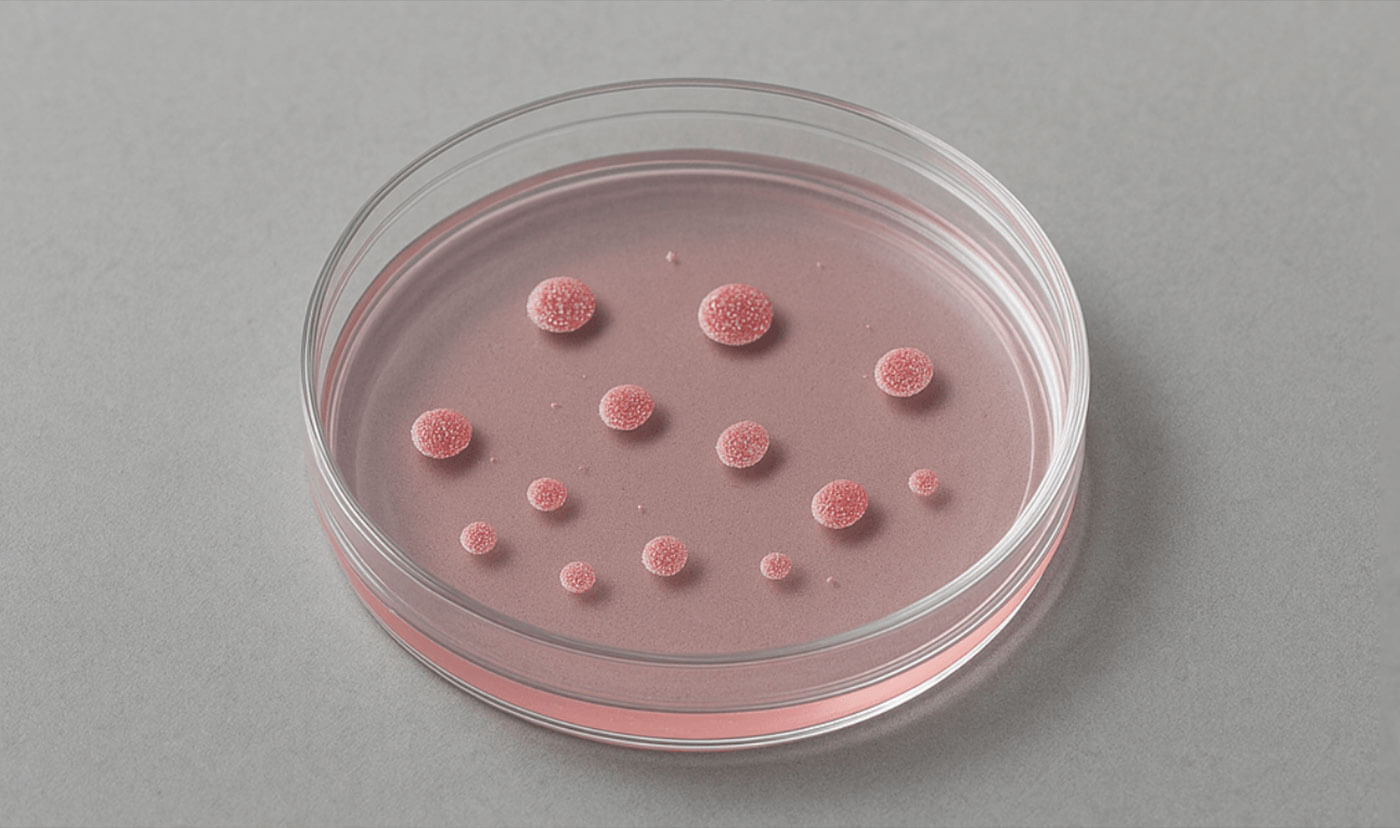
Lab-in-the-loop
Before clinical trials, new treatments must be tested in model laboratory systems. Owkin’s lab-in-the-loop specializes in a state-of-the-art model system: patient-derived organoids.
These are mini versions of human organs, grown in petri dishes from patient cells. They match what’s happening in the human body more closely than other lab model systems, so the results are more likely to be accurate.
Owkin's AI Scientist
We're building thousands of AI scientists that work 24/7 to accelerate medical breakthroughs, to compress decades of discovery into years.
The AI agent for biology, powered by multimodal patient data, for smarter biopharma decision making.

